 
Mole × molecular weight = mass (rearrange the equation),Īs 1.626×10 23 molecules of NaOH is also equal to 0.27 moles, and we know that the molecular weight of NaOH is 40, we can use these numbers to get: Mole = mass / molecular weight (multiply both sides by molecular weight), As we need to find the mass of NaOH to add, let's quickly rearrange the equation: Now we know the number of molecules of HCl we have, and since the reaction is 1:1, we need the exact same number of molecules of NaOH to neutralize it. We can work out the number of molecules by multiplying the moles by Avogadro's constant above. Let's plug these numbers into the above equation: 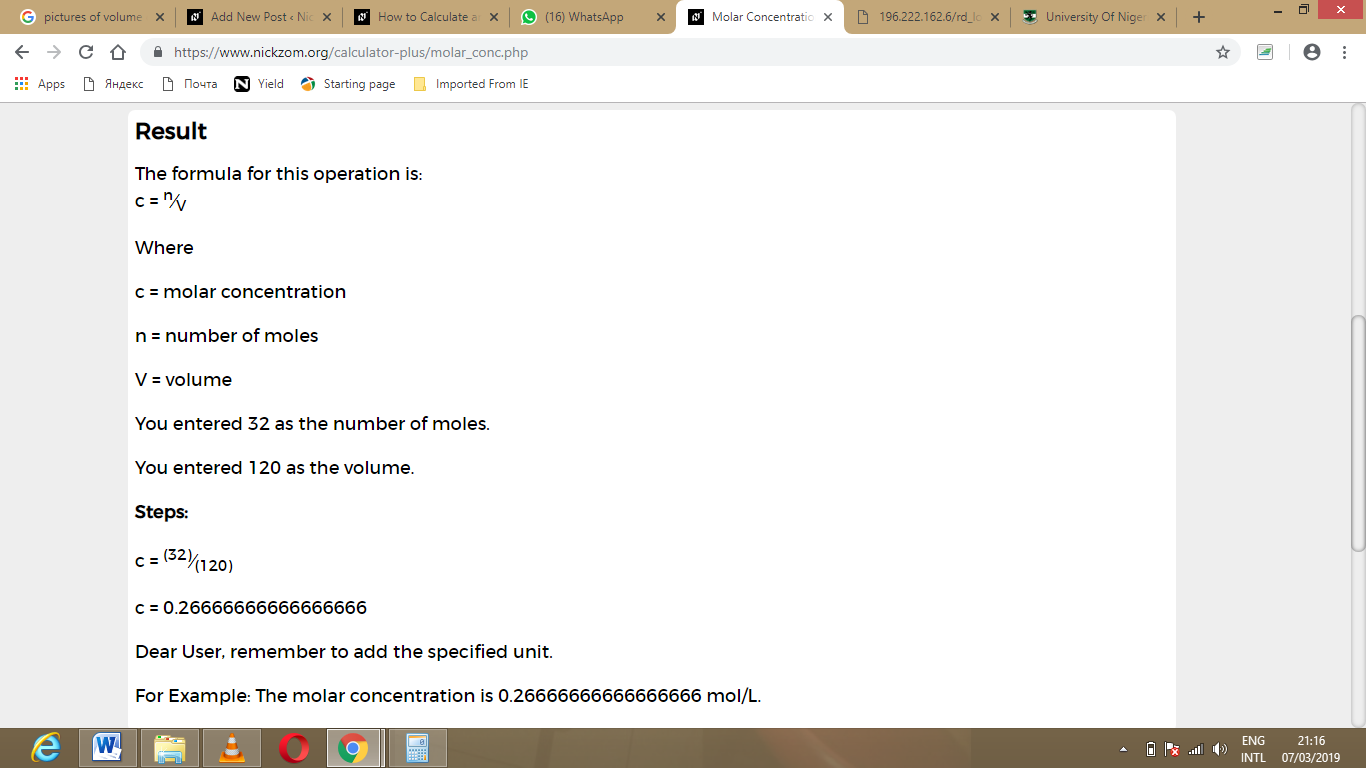
We know we have 10 g of HCl, which has a molecular weight of 36.5 g/mol. Let's do a quick example to help explain how to convert from moles to grams or grams to moles. If you wanted to find the concentration of the hydrochloric acid, you could use our concentration calculator. 
To know how to calculate moles, the equation is: It is, therefore, useful to find out exactly how many molecules of HCl are in the solution. Now, you want the resulting solution to be perfectly neutral, so you don't want to add too much or too little NaOH, making it too basic or acidic respectively. Let's say you want to neutralise 10 g of hydrochloric acid (HCl in water) with some sodium hydroxide (NaOH). So why do chemists use moles, and why do you need a mole calculator? Well, as we said above, it provides a useful metric when dealing with reactions. It turns out memorising that definition for my GCSE chemistry exam was pointless. Prior to that, a mole was defined as the number of atoms in 12 grams of carbon-12 (an isotope of carbon). The official International System of Units definition is that a mole is the amount of a chemical substance that contains exactly 6.02214076×10 23 ( Avogadro's constant) atoms, molecules, ions or electrons (constitutive particles), as of 20 th May 2019. A mole is how chemists define an amount of substance, useful when dealing with many different molecules reacting at once (i.e., any reaction). Just kidding – we're sure you've never heard that joke before. Last accessed: 29 August 2020 ( paid link).A mole is a small, subterranean mammal belonging to the family Talpidae. Published by McGraw-Hill Professional 1 edition (November 20, 2002). Handbook of Inorganic Chemicals by Pradyot Patnaik (Author). Last accessed: 29 August 2020 (.gov/compound). National Library of Medicine 8600 Rockville Pike Bethesda, MD 20894 USA. National Center for Biotechnology Information U.S. 
Compound summary: Calcium chloride dihydrate. Calcium chloride monohydrate weighs 2 240 kg/m³ (139.83863 lb/ft³).Calcium chloride hexahydrate weighs 1 710 kg/m³ (106.75181 lb/ft³).Calcium chloride anhydrous weighs 2 150 kg/m³ (134.22012 lb/ft³).A few materials, substances, compounds or elements with a name containing, like or similar to Calcium chloride dihydrate:.Also known as: Calcium dichloride dihydrate.Įlements: Calcium (Ca), Chlorine (Cl), Hydrogen (H), Oxygen (O).In Imperial or US customary measurement system, the density is equal to 115.492 pound per cubic foot, or 1.069 ounce per cubic inch. density of calcium chloride dihydrate is equal to 1 850 kg/m³. Calcium chloride dihydrate weighs 1.85 gram per cubic centimeter or 1 850 kilogram per cubic meter, i.e. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
